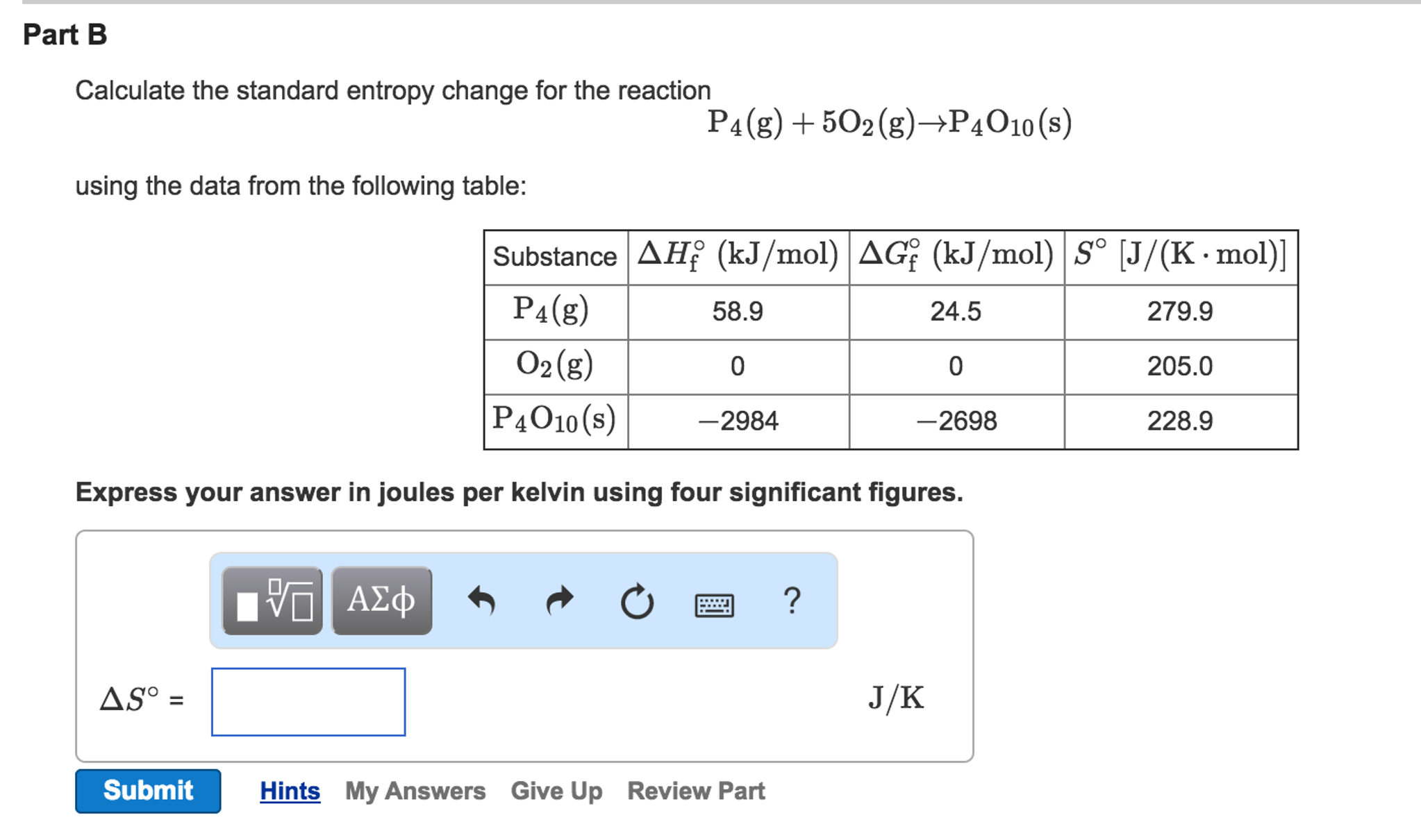
In other words, the maximum efficiency is achieved if and only if entropy does not change per cycle. In the real world, this may be difficult to achieve since the cold reservoir is often an existing ambient temperature.  
Looking at this formula an interesting fact becomes apparent: Lowering the temperature of the cold reservoir will have more effect on the ceiling efficiency of a heat engine than raising the temperature of the hot reservoir by the same amount. Rearranging the right side of the equation gives what may be a more easily understood form of the equation, namely that the theoretical maximum efficiency of a heat engine equals the difference in temperature between the hot and cold reservoir divided by the absolute temperature of the hot reservoir. A corollary to Carnot's theorem states that: All reversible engines operating between the same heat reservoirs are equally efficient. Step 3: Work out the entropy change using the formula S. Thus, Equation 3 gives the maximum efficiency possible for any engine using the corresponding temperatures. This enthalpy change cannot be measured directly so a Born-Haber cycle is used to determine. Irreversible systems and losses of energy (for example, work due to friction and heat losses) prevent the ideal from taking place at every step.Ĭarnot's theorem is a formal statement of this fact: No engine operating between two heat reservoirs can be more efficient than a Carnot engine operating between those same reservoirs. For this figure, the curve indicates a vapor-liquid equilibrium ( See Rankine cycle). This change is indicated by the curve on a T– S diagram. The entropy of a real material changes with temperature. In a Carnot cycle, a system or engine transfers energy in the form of heat between two thermal reservoirs at temperatures T H, none can exceed the efficiency of a Carnot cycle.įigure 6: A real engine (left) compared to the Carnot cycle (right). dQ de + p dV Using the equation of state, we can. The equations can be formulated in terms of the internal energy and the definition of work for a gas. For gases, there are two possible ways to evaluate the change in entropy. By Carnot's theorem, it provides an upper limit on the efficiency of any classical thermodynamic engine during the conversion of heat into work, or conversely, the efficiency of a refrigeration system in creating a temperature difference through the application of work to the system. The change in entropy is then the inverse of the temperature integrated over the change in heat transfer. A Carnot cycle is an ideal thermodynamic cycle proposed by French physicist Sadi Carnot in 1824 and expanded upon by others in the 1830s and 1840s.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
